Shellfish Aquaculture Program
In partnership with local governmental and private agencies, farmers, and researchers, Roger Williams University's shellfish program implements research methods to inform regional strategies for restoration and management of quahog and oyster populations.
We test hatchery and grow-out technologies for commercially significant species, like oysters and quahogs, and we are working to establish methods new species for commercial production, to expand aquaculture opportunities in the region and promote industry growth and job creation.
From Hatchery to Harvest
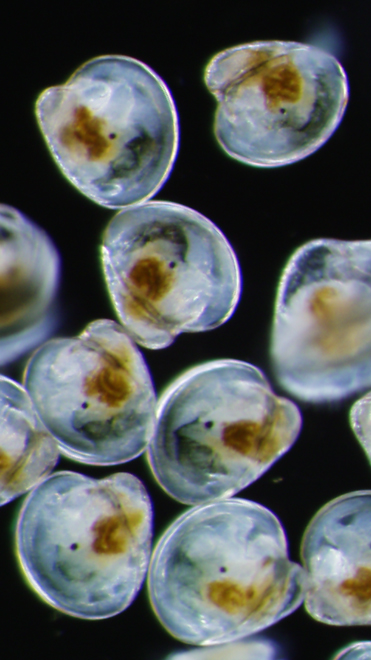
As part of our mission to support the region’s blue economy, our Shellfish Program focuses on education, research, partnerships, and innovation that support sustainable aquaculture - use of ocean resources for economic growth, improved livelihoods, and ocean health.
The Shellfish Program at Roger Williams University consists of all three phases of shellfish aquaculture; hatchery, nursery, and grow-out. The program through its partnerships also assists the wild fisheries in research for bivalves native to Rhode Island waters. Through the Shellfish Hatchery, the FerryCliffe Shellfish Farm, and field research, our faculty and staff engage with the industry as an extension resource.
Student Opportunities
Our spaces serve as a place for student training and as a production facility for regional shellfish studies. CEED’s Shellfish Program offers undergraduate students the unique opportunity to engage in all aspects and phases of shellfish aquaculture – from hatchery to harvest.
RWU students can join the Shellfish Program to gain aquaculture work experience, to conduct applied research in support of the programming, or both.
Students learn microalgal culture, shellfish hatchery operation, nursery development, farm management, and they can collaborate on regional restoration initiatives, work with local shellfish farmers, researchers, and in the community. This industry-relevant experience in shellfish aquaculture research and practice prepares our students as the next generation of the region’s skilled workforce for a thriving Blue Economy.
One of our well-known training programs available to both RWU students and the public is the Applied Shellfish Farming course, which is offered annually by both the RWU Shellfish Hatchery Manager and the RWU Shellfish Field Technician. This course will provide interested individuals with the technical information needed to confidently undertake a shellfish farming enterprise. All aspects of shellfish farming, from the broodstock to the market, will be covered over the fifteen-week course. Participants will learn the basic principles of hatchery, nursery and grow-out operations; as well as risk management, siting and permitting, and business management.
Check out our updates on Instagram at @RWU_Shellfish_Program
Microalgae Production: schamberlin@rwu.edu
Facilities
RWU has aquaculture facilities on campus - a unique asset for a university - providing opportunities for our students, staff and faculty to lead the way in aquaculture education, research, training, and innovation.
 CEED operates one of the three shellfish hatcheries in Rhode Island, but it is the only one within a university, and it produces shellfish seed for a variety of species and projects, including restoration and academic research. Established in 2004, this facility was first located in the wet lab, and by 2010, a 4000 ft2 expansion of the MNS Building established the Luther H. Blount Shellfish Hatchery -- a larger facility designed to allow dedicate activity in our focus areas: Research, Education & Training, Extension & Outreach, Restoration, and Commercial Sales. The CEED Luther H. Blount Shellfish Hatchery has the capacity to provide aquaculture training for our students and to the general public. The hatchery primarily produces shellfish for projects in Rhode Island, but also for wider collaborations along the east coast of the United States.
CEED operates one of the three shellfish hatcheries in Rhode Island, but it is the only one within a university, and it produces shellfish seed for a variety of species and projects, including restoration and academic research. Established in 2004, this facility was first located in the wet lab, and by 2010, a 4000 ft2 expansion of the MNS Building established the Luther H. Blount Shellfish Hatchery -- a larger facility designed to allow dedicate activity in our focus areas: Research, Education & Training, Extension & Outreach, Restoration, and Commercial Sales. The CEED Luther H. Blount Shellfish Hatchery has the capacity to provide aquaculture training for our students and to the general public. The hatchery primarily produces shellfish for projects in Rhode Island, but also for wider collaborations along the east coast of the United States.
Here is a list of commonly cultured invertebrate species in the hatchery:
Crassostrea virginica - Eastern oyster
Mercenaria mercenaria - Hard clam
Argopecten irradians - Bay scallop
Ensis directus - Atlantic razor clam
Tagelus plebeius - Stout razor clam
Mya arenaria - Soft shell clam
Mytilus edulis - Blue mussel
Anadara granosa - Blood ark
Solemya velum - Atlantic awning clam
The hatchery also produces kelp spools of Sugar Kelp (Saccharina latissima), with other species available. Seaweed aquaculture is a growing area of interest for the industry. Currently, the primary use of kelp in the Northeast is a way to diversify a farm's crop in the off-season or in Integrated Multi-Trophic Aquaculture (IMTA), but as the market improves for this product, it can possibly be a stand alone crop for a farmer. The micro- and macro-algal production has the same areas of focus as the shellfish production. RWU undergraduate students are incorporated in all phases of hatchery activities.
Since algae is the food for all stages of shellfish development, the most important part of a shellfish hatchery is its algal culture. In fact, algal production for a shellfish hatchery is the largest proportion of the total operational expense, and can account for about 30-50% of a shellfish hatchery’s costs. At CEED we produce different mixes of algae species for different life stages to both promote development and meet food size requirements at different developmental stages of shellfish.
The Shellfish Hatchery collaborates with the Sharp Lab’s Temperate Corals and Microbial Ecology research program to support efforts for spawning and breeding the temperate coral Astrangia poculata in captivity.

Student Assistants: Each year, between 15-20 students help operate hatchery and conduct specific shellfish research projects.
 CEED has operated and maintained the FerryCliffe Shellfish Farm at RWU since 2010. The farm consists of a variety of aquaculture gear including both on bottom and floating gear as well as an operational upweller system. Our staff and faculty conduct a variety of field research on partner shellfish farms and field sites around Rhode Island. The focus areas for the farm are Research, Education & Training, Extension & Outreach, and Restoration. Through collaborative research, we develop new shellfish culture technologies from the nursery to market stage. Through hands-on experience, students and the general public have the opportunity to learn all aspects of shellfish cultivation on a working aquaculture farm. With the help of our students, we produce FerryCliffe oysters which are donated to university functions and local non-profit events.
CEED has operated and maintained the FerryCliffe Shellfish Farm at RWU since 2010. The farm consists of a variety of aquaculture gear including both on bottom and floating gear as well as an operational upweller system. Our staff and faculty conduct a variety of field research on partner shellfish farms and field sites around Rhode Island. The focus areas for the farm are Research, Education & Training, Extension & Outreach, and Restoration. Through collaborative research, we develop new shellfish culture technologies from the nursery to market stage. Through hands-on experience, students and the general public have the opportunity to learn all aspects of shellfish cultivation on a working aquaculture farm. With the help of our students, we produce FerryCliffe oysters which are donated to university functions and local non-profit events.
Aquaculture Field Operations Manager:Malcolm Bowen (mbowen@rwu.edu)
Student Assistants: Most field research is conducted during the summer months and is based on grant funding. Typically 2-8 students are hired over the summer to help with field projects based on external funding. Student research credit (AQS 450) opportunities, however, are available during the fall and spring semesters. Please contact Malcolm Bowen (mbowen@rwu.edu) about these potential opportunities.
Projects & Partnerships
Current Projects
RIDEM-RWU-RISA Quahog Seeding Long-term Collaborative Partnership (2005-present)
For many years, our shellfish program has collaborated with the Rhode Island Shellfisherman’s Association (RISA) and RI Department of Environmental Management (DEM) to raise oyster and quahog seed in upwellers run by RISA members during the summer and fall months. RISA members then seed the young bivalves in November. In recent years we have focused on quahogs specifically. The RWU shellfish team and students collaborate by providing seed and collecting growth rate data every year.
Partners: RI Shellfisherman’s Association (RISA) and RI DEM (funder)
Warren Oyster Enhancement (2005-present)
For the last 15 years, faculty and staff of the RWU Shellfish Program have collaborated with the town of Warren, RI to enhance oyster production in an area where local residents recreationally harvest. In 2021 we received additional funds from the Shellfish Restoration Foundation of Narragansett Bay to expand the project to explore different kinds of grow out methods for oysters in Warren’s recreational harvest area.
Partners: Town of Warren, Hassenfeld Community Project (Funder), Saltbox Sea Farm, Shellfish Restoration Foundation of Narragansett Bay (Funder)
Temporal and spatial dynamics of rust tide caused by Margalefidinium polykrikoides (Mp) on Rhode Island shellfish farms in Narragansett Bay and resulting impacts on cultured oysters (2022-2024)
Rust tide is a type of Harmful Algal Bloom that can cause mortality in fish, crustaceans and bivalves, but is not toxic to humans. In recent years, these blooms have increased in frequency in Rhode Island, causing significant harm to oyster farms in the state.
Our two primary of this project – a) determining spatio-temporal dynamics of rust tide by applying environmental DNA methods, and b) collecting baseline data on direct impacts of Mp on the most vulnerable stages of oysters – will be critical to local mitigation strategies against rust tide events in Rhode Island.
The research objectives of our proposal also align with Rhode Island Sea Grants 2018-2023 goals to support healthy coastal ecosystems, promote sustainable fisheries and aquaculture and develop resilient communities and economies in Rhode Island.
Partners: NOAA NEFSC Milford Laboratory, Behan Family Farms, Rhode Island Sea Grant (funder)
Identification of hard clams resistant to HN disease and development of diagnostic test methods (2022-2023)
Hemocytic Neoplasia (HN) is causing significant mortality in Mercenaria mercenaria (hard clam) aquaculture in Massachusetts and has been identified in hard clam stocks in other states including Rhode Island, New Jersey, and Florida. Results from yearly monitoring and recent research demonstrates that, while the disease is contagious and appears to infect all strains of hard clams, there is a genetic basis for resistance to the disease. Thus, development of resistant hard clam strains is a possible solution to the devastating effects of HN in cultured hard clams. The primary objectives of the study are to 1) develop hard clams resistant to HN, 2) identify of genes that contribute to infection/disease resistance, 3) develop a molecular diagnostic test method, 4) provide potentially resistant broodstock to hatcheries and molecular diagnostic tests to shellfish laboratories and 5) provide information from this study to culturists and regulators to use in managementdecisions.
Our hatchery is helping produce four of six lineages to support the project with the Aquatic Diagnostic Laboratory as lead.
Partners: RWU’s Aquatic Diagnostic Laboratory (lead), University of Rhode Island, USDA, Cape Cod Cooperative Extension, A.R.C., Northeastern Regional Aquaculture Center (NRAC) (funder)
Bottle Upweller System (2021- 2023)
The goal of this project is to optimize the performance of the Bottle UPweller SYstem (BUPSY) in supporting juvenile shellfish growth. To meet that goal, we will complete the following goals:
- Characterize the flow dynamics of various shapes and sizes of commonly used bottle upwellers. To characterize these dynamics, we will conduct dye tracer studies and create computational fluid dynamic models for these vessels.
- Determine the role that both vessel shape and water velocity through the BUPSY play in enhancing the overall production of post-set juvenile shellfish in the system.
- Using the results from the above two goals, we will develop and evaluate an optimized BUPSY design that we will test in two commercial hatcheries.
We will share these results with commercial shellfish hatcheries and other interested parties. By providing shellfish hatcheries with an optimized BUPSY, the production time for saleable seed coming from the hatchery will be reduced, resulting in the potential for producing more seed and holding it for shorter time intervals in the hatchery.
Partners: Industry partners: Aquaculture Research Corp. and Aeros Cultured Oyster Co., Northeastern Regional Aquaculture Center (NRAC) (funder)
Impact of Biofouling on Rhode Island Aquaculture Farms (2021-2022)
Biofouling organisms can be particularly damaging to aquaculture business success. Too much fouling on oyster culture gear can slow water flow, reducing the availability of oxygen, food supply, and the ability to remove waste. Ultimately this can reduce the growth rates of oysters or cause them to die. This project focuses on estimating the quantity of biofouling organisms on gear at farms around Rhode Island.
Partners: Coastal Resource Management Council, Saltbox Sea Farm, US Department of Interior (Funder)
Side Effects of Breeding Disease-Tolerant Oysters (2020-2022)
Commercial oyster seed is limited to specific environments and vulnerable to naturally-occurring diseases. Despite its growing value both economically and environmentally, northeastern U.S. oyster production is constrained by the lack of commercial seed available for specific growing environments and losses from diseases. Furthermore, changes in climate are expected to augment existing constraints even further. This project will investigate whether oyster hatcheries and selective breeding programs select for disease resistance or tolerance, and what that may mean for disease dynamics in coastal marine ecosystems. Our parallel approach integrating targeted laboratory experiments, data collected through mesocosm experiments, and dynamic models will improve our capacity to predict and forecast host-parasite dynamics and its consequences for oysters. Results will also identify oyster stocks with the best potential for the development of lines with improved performance in the Northeast region both for aquaculture production and restoration. RWU’s Hatchery Manager Rob Hudson is responsible for the spawning and raising of all oyster seed in this project.
Partners: University of Rhode Island, RI Sea Grant (funder), United States Department of Agriculture: Agricultural Research Service (USDA ARS), and North Carolina State University.
Iceland Scallop Population Dynamics (2021-2022)
The Iceland scallop fishery ultimately collapsed in 2004 in Iceland due to the combined effects of overfishing, recruitment failure, and disease. The remaining populations have been surveyed most years since then by the Hafronnsóknastofnun (Marine and Freshwater Research Institute) in Iceland. This is a long-term dataset of spat fall, gonad condition, recruitment and stock of Iceland scallops. Dr. Bayer, in collaboration with a colleague at MFRI are analyzing these data for temporal patterns of population dynamics, examining the effects of both environmental and internal population factors
Partners: Hafrannsóknastofnun (Marine and Freshwater Research Institute), Iceland Fulbright Commission (funder)
Nitrogen Extraction by Oyster Farms (2019-2022)
Oysters are incredibly effective filters and possess the capability of improving water quality fairly quickly. Often in coastal systems we have the problem of excess nitrogen input from human activities. However, oysters have the potential to reduce this excess fertilizer (nitrogen) from the water through filtration. They absorb it into their bodies as they grow as well as draw it down into the sediments through feces and pseudofeces. In collaboration with Stella Mar Oysters and the computer modeling firm Longline Environment, Dr. Bayer and scientists at NOAA Milford Lab have investigated how much nitrogen oysters at one farm in Connecticut can extract out of the water column. These data will be helpful for determining how policy-makers can credit oyster farmers for the filtration of nitrogen done by their oysters.
Partners: NOAA Milford Lab, Stella Mar, Longline Environment, NOAA Office of Aquaculture (funder)
Addressing Shell Fragility In Shellfish Nursery Systems
 Shell fragility is a common problem in shellfish nursery systems worldwide. As such, the development of a calcium-based product that will improve shell strength in nursery reared shellfish of many different bivalve species then a global market could open up. Shellfish hatcheries are becoming more and more common in replacing wild caught shellfish spat for supplying shellfish aquaculture internationally.
Shell fragility is a common problem in shellfish nursery systems worldwide. As such, the development of a calcium-based product that will improve shell strength in nursery reared shellfish of many different bivalve species then a global market could open up. Shellfish hatcheries are becoming more and more common in replacing wild caught shellfish spat for supplying shellfish aquaculture internationally.
Partners: Full Measure Industries, LLC, RI Commerce The Innovation Voucher Program (funder).
Resilience To Climate Change: Testing Sculptural Forms for Coastal Habitat Restoration
Successful implementation of habitat restoration projects that minimize coastal hazards and increase the resilience of coastal regions to the impacts of global climate change requires the integration of scientific knowledge about coastal ecosystems with an understanding of the economic, social, and cultural factors affecting governance and decision-making. A key element of this process is public engagement. The goal of this project was to develop and implement a coastal habitat restoration project at an urban site in Rhode Island that directly engaged the public into research on the impact of climate change on coastal ecosystems.
Partners: University of Rhode Island, Rhode Island School of Design (RISD), Rhode Island College (RIC), RI Research Alliance Collaborative Grants Program (funder).
Freshwater Aquaculture
Our freshwater projects are mainly used for student research, but have great potential for aquaculture industry development. Projects include freshwater ornamental aquatic plant cultivation (hydroponics), koi production and the development of freshwater fish (largemouth bass) culture in Rhode Island.
Cranberry Bog Conversion
In 2002, in response to a weak market for locally grown cranberries, Dale Leavitt worked with a local producer from Ocean Spray to convert a cranberry bog to fish production. This provides an alternative crop while keeping the land in productive use and the pond can quickly revert to cranberry production when the market improves. Fish were successfully and profitably produced for several years until the cost of diesel fuel began to rise. As the pond lacks direct access to utilities, electricity to run the circulation pumps had come from a diesel generator. In 2008, Dr. Leavitt received a planning grant from the EPA that, working cooperatively with the RWU School of Engineering, funded the design of a new pond using solar panels, an alternative and sustainable energy source, to run the pumps. A second EPA grant led to the construction of this new pond in 2010.
Solar UpwellerSolar Upweller
Funded through a USDA grant, Dale Leavitt planned and constructed a solar powered FLUPSY – a floating dock/upweller system used for the cultivation of shellfish. This is state-of-the-art for shellfish nursery culture, but currently FLUPSY's must be at a marina so they can be hooked up to a power source. Dr. Leavitt and his team of students from Marine Biology and the School of Engineering made significant improvements to the FLUPSY design so that a smaller pump utilizing less power can be used. In addition, they added solar panels, a battery bank and a back-up system. The proof of concept for this project occurred on campus during fall, 2008 and the unit was first deployed in the field in summer, 2009.
Hydroponics
Hydroponics is the science of growing vegetables/herbs without soil. In the past, we have combined hydroponic production with fish cultivation in a single system – the fish provide nutrients for the plants while the plants, in turn, clean up the water for the fish. Both fish and vegetables can be grown and harvested from systems such as these. Effluent environmental management is an important component of fish farming, and plants harvested with the root intact tend to have a longer shelf life.
New England Marine Ecology Marine Economy
There is a growing recognition of the diversity, importance and impact of the marine industrial sector to the general economic health of the region. Narragansett Bay is often cited as an economic engine for Rhode Island, but the economic impact is not widely recognized. One attempt to summarize this impact was during the 2-day "Bay Summit" hosted by the DEM in 2000. RWU was greatly involved in that effort, but the conference intent of continued discussion and nurturing was never realized. In April 2007, RWU and the New England Council hosted a one day conference to further explore this topic.
Reef Balls
In 2006, we established the first reef ball project in Rhode Island. This emerging technology is being used to establish oyster reefs while the three dimensional structure provides habitat for juvenile fish. The project was funded by a small grant through the Nature Conservancy to David Taylor, and the work is being done collaboratively with the CEED hatchery.
Oyster Disease Resistance
This is a long-term project involving Dale Leavitt and Roxanna Smolowitz in collaboration with Marta Gomez-Chiarri of URI to develop and deploy a local strain of oysters resistant to common diseases.
The highest risk for both wild harvest oysters and oyster farming is disease. Parasitic diseases such as Dermo and MSX have devastated the American oyster industry in the US, resulting in millions of dollars of losses. To counter the impact of disease, strains of oysters resistant to Dermo, MSX, and Juvenile Oyster Disease have been developed through selective breeding. Few of these strains have a Northeast origin and none have been tested in local growing conditions. Thanks to funding through the RI Aquaculture Initiative and the USDA-SARE Program, we have been testing the performance of two putative disease resistant oyster strains (NEH and FMF), and a local RI strain (GHP) at local aquaculture farms.
The second phase of this project, currently underway, is to develop a local line of oysters with disease resistance that will be adapted to local growing conditions. This was accomplished by performing crosses of local oysters with disease resistant strains. The offspring are now being evaluated in the field (a multi-year process) and we anticipate the results being extremely valuable to farmers so they can make informed decisions regarding strain choice.
Impact of Clean Water on Quahog Production
The Narragansett Bay Commission, which operates the major sewage treatment plants for Rhode Island, recently opened the first phase of the combined sewer outfall (CSO) pollution abatement system – a planned series of tunnels that will collect waste water during rain storms and hold it until it can be properly treated. This is a common, if expensive, solution to the problems of CSO’s and it’s anticipated that this will result in cleaner water in the upper bay. As this might result in the opening of waters long-closed to commercial shellfishing, we are working on an assessment of how this could potentially impact quahog larval supply and distribution, as well as an analysis of production and market impacts if quahog landings from the upper bay increase.
International Projects
- Clam Aquaculture in China
- Marine Ornamental Production in Sri Lanka
- Aquaculture in Turkey
Shellfish Partners
The CEED Shellfish Program has had a number of excellent partner organizations:
- Coastal Resource Management Council
- RI Department of Environmental Management
- Narragansett Bay Commission
- Rhode Island Shellfisherman’s Association
- University of Rhode Island
- NOAA Milford Lab
- United States Department of Agriculture:
- Agricultural Research Service (ARS)
- Animal and Plant Health Inspection Service (APHIS)
- The Natural Resources Conservation Service (NRCS)
- The Town of Warren, RI
- Shellfish Restoration Foundation of Narragansett Bay
- The Commercial Fisheries Research Foundation (CFRF)
- RI Sea Grant
- Save The Bay
- The Nature Conservancy