ADL Research and Publications
Research
The ADL engages in externally funded aquatic animal health research and trains RWU students in aquatic animal health diagnostics and care methods.
Current Areas of Research
- Health and resilience of aquatic organisms in a changing environment
- Development of new diagnostic tests is an important goal in the laboratory. Molecular detection and quantification test methods currently under development include
- PCR and qPCRs for protozoan parasites occurring in sea scallops
- A multiplex qPCR for important parasites causing morbidity and mortality in marine aquarium fishes
- Pathogen characterization and disease etiology
- One of our main focuses is on the occurrence of neoplasia in hard clams. We have determined that the disease is spread directly, most likely by a neoplastic cell, and is affecting populations of aquacultured hard clams in the northeast. We are currently evaluating various hard clam populations to determine the extent of the disease in MA and surrounding states and are working with regulatory agencies to determine how to best address the problems caused by this new hard clam disease.
- Environment-Host-Microbiome interactions
- Microbiomes of sea turtles that are stranded vs. those in the oceans
- Environment DNA (eDNA) detection tools

Hemocytic Neoplasia
Our current research involves the epidemiology of, and the molecular basis for, the occurrence of neoplasia in hard clams (funded by Northeast Regional Aquaculture Center and the USDA). This disease was first identified in Mercenaria mercenaria (hard clams) in MA.
Sea Turtle Microbiomes
Determination of the microbiome of sea turtles is a recent focus of research (with the Coonamessett Farm Foundation). Identifying the microbiome of sea turtles that are stranded vs. those in the oceans, may help in developing effective stranding treatment methods.
New Diagnsotics
Developing new diagnostic tests is an important goal in the laboratory. Molecular detection and quantification test methods currently under development include tests for an important protozoan parasite occurring in sea scallop muscles, and a test for three important protozoan parasites causing morbidity and mortality in marine aquarium fishes.
Publications
Journal Articles
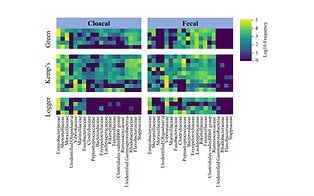
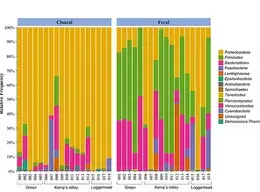
Forbes ZR, Scro AK, Patel SH, Dourdeville KM, Prescott RL, Smolowitz RM. 2023.Fecal and cloacal microbiomes of cold-stunned loggerhead Caretta caretta, Kemp’s ridley Lepidochelys kempii, and green sea turtles Chelonia mydas. Endangered Species Research. https://doi.org/10.3354/esr01220
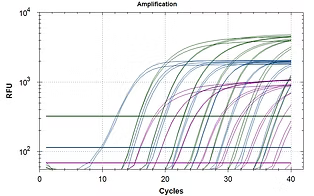
Piesz JL, Scro AK, Corbett R, Lundgren KM, Smolowitz R, Gomez-Chiarri M. 2022. Development of a multiplex qPCR for the quantification of three protozoan parasites of the eastern oyster, Crassostrea virginica. Diseases of Aquatic Organisms. https://doi.org/10.3354/dao03694
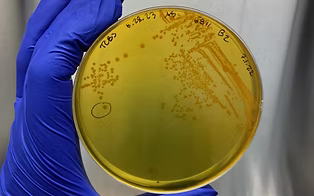
Scro AK, Westphalen J, Kite-Powell HL, Brawley JW, Smolowitz RM. 2022. The effect of off-bottom versus on-bottom oyster culture on total and pathogenic Vibrio spp. abundances in oyster tissue, water and sediment samples. International Journal of Food Microbiology. https://doi.org/10.1016/j.ijfoodmicro.2022.109870
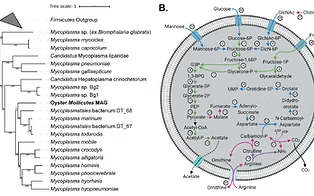
Pimentel ZT, Dufault-Thompson K, Russo KT, Scro AK, Smolowitz RM, Gomez-Chiarri M, Zhang Y. 2021. Microbiome analysis reveals diversity and function of Mollicutes associated with the eastern oyster, Crassostrea virginica. mSphere. https://doi.org/10.1128/mSphere.00227-21
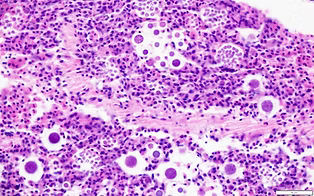
Scro AK, Lundgren KM, and Smolowitz R. 2019. Multiplex qPCR detection of non-pathogenic and pathogenic Vibrio parahaemolyticus and Vibrio vulnificus from oysters cultured in Massachusetts and Rhode Island: the reliability of MPN as an indicator. Journal of Shellfish Research. https://doi.org/10.2983/035.038.0112

Smolowitz R. 2018. A Review of QPX Disease In The northern quahog(= Hard Clam) Mercenaria mercenaria.Journal of Shellfish Research 37: 807-819. https://doi.org/10.2983/035.037.0411
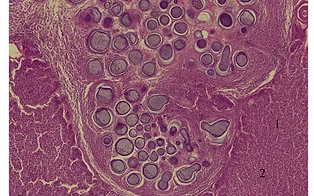
Huntsberger CJ, Hamlin JR, Smolowitz RJ, Smolowitz RM. 2017. Prevalence and description of Ichthyophonus sp. in yellowtail flounder (Limanda ferruginea) from a seasonal survey on Georges Bank. Fisheries Research 194: 60-67. https://doi.org/10.1016/j.fishres.2017.05.012
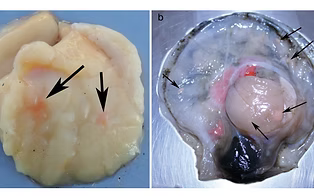
Grimm C, Huntsberger C, Markey K, Inlgis S, Smolowitz R. 2016. Identification of a Mycobacterium sp. as the causative agent of orange nodular lesions in the Atlantic sea scallop Placopecten magellanicus. Disease of Aquatic Organisms 118: 247-258. https://doi.org/10.3354/dao02961

Sohn S, Lundgren KM, Tammi K, Smolowitz R, Nelson DR, Rowley DC, Gómez-Chiarri M. 2016. Efficacy of Probiotics in Preventing Vibriosis in the Larviculture of Different Species of Bivalve Shellfish. Journal of Shellfish Research 35: 319-328. https://doi.org/10.2983/035.035.0206

Sohn S, Lundgren KM, Tammi K, Karim M, Smolowitz R, Nelson DR, Rowley DC, Gómez-Chiarri M. 2016. Probiotic Strains for Disease Management in Hatchery Larviculture of the Eastern Oyster Crassostrea virginica. Journal of Shellfish Research 35: 307-317. https://doi.org/10.2983/035.035.0205
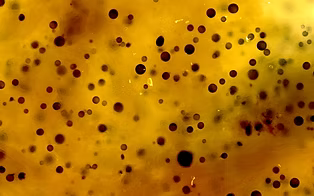
Smolowitz R. 2013. A Review of Current State of Knowledge Concerning Perkinsus marinus Effects on Crassostrea virginica (Gmelin) (the Eastern Oyster). Veterinary Pathology 50: 404-411. https://doi.org/10.1177/0300985813480806

De Faveri J, Smolowitz RM, Roberts SB. 2009. Development and Validation of a Real-Time Quantitative PCR Assay for the Detection and Quantification of Perkinsus marinus in the Eastern Oyster, Crassostrea virginica. Journal of Shellfish Research 28: 459-464. https://doi.org/10.2983/035.028.0306
Book Chapters
Smolowitz R. 2021. Mollusca: Bivalvia. In: LaDouceur EEB, ed. Invertebrate Histology. Hoboken, NJ: John Wiley 163–183. https://doi.org/10.1002/9781119507697.ch6
Getchell RG, Smolowitz RM, McGladdery SE, Bower SM. 2016. Chapter 10 - diseases and parasites of scallops, in: Shumway SE, Parsons GJ (Eds.). Developments in Aquaculture and Fisheries Science. Elsevier 425–467. https://doi.org/10.1016/B978-0-444-62710-0.00010-9